From Self-Reflection to a Research Path
After completing her bachelor’s degree in Illinois Tech in Chicago and returning home to Malaysia, Nazia Aslam began thinking seriously about what she wanted for her future. Although she had always been interested in science, pursuing a PhD was not something she had previously considered or seen people around her do.
“I started asking myself what kind of life I truly wanted, not just what was available to me, but what I could build for myself,” Nazia said. “I realized that I wanted to do meaningful work, to challenge myself, and to create opportunities that didn’t yet exist in my world.”
That realization led her back to the United States, where she is now a fourth-year Chemical Engineering PhD student at the University of Connecticut. As the first person in her family to pursue this path, Nazia describes the decision as both difficult and transformative.
Process Intensification of Water-Treatment Systems
Today, her research bridges chemical engineering, machine learning, and water treatment systems. Her work focuses on developing interpretable and efficient models that can uncover governing physics and improve the design and operation of complex dynamic processes.
At the beginning of her PhD, Nazia worked on comparative studies involving physics-informed neural networks and symbolic regression to identify governing partial differential equations from sparse and noisy data. Her work was presented at venues including a Data-Driven Physical Simulations seminar at Lawrence Livermore National Laboratory, a CRUNCH group webinar at Brown University, and the 2023 INFORMS Annual Meeting.
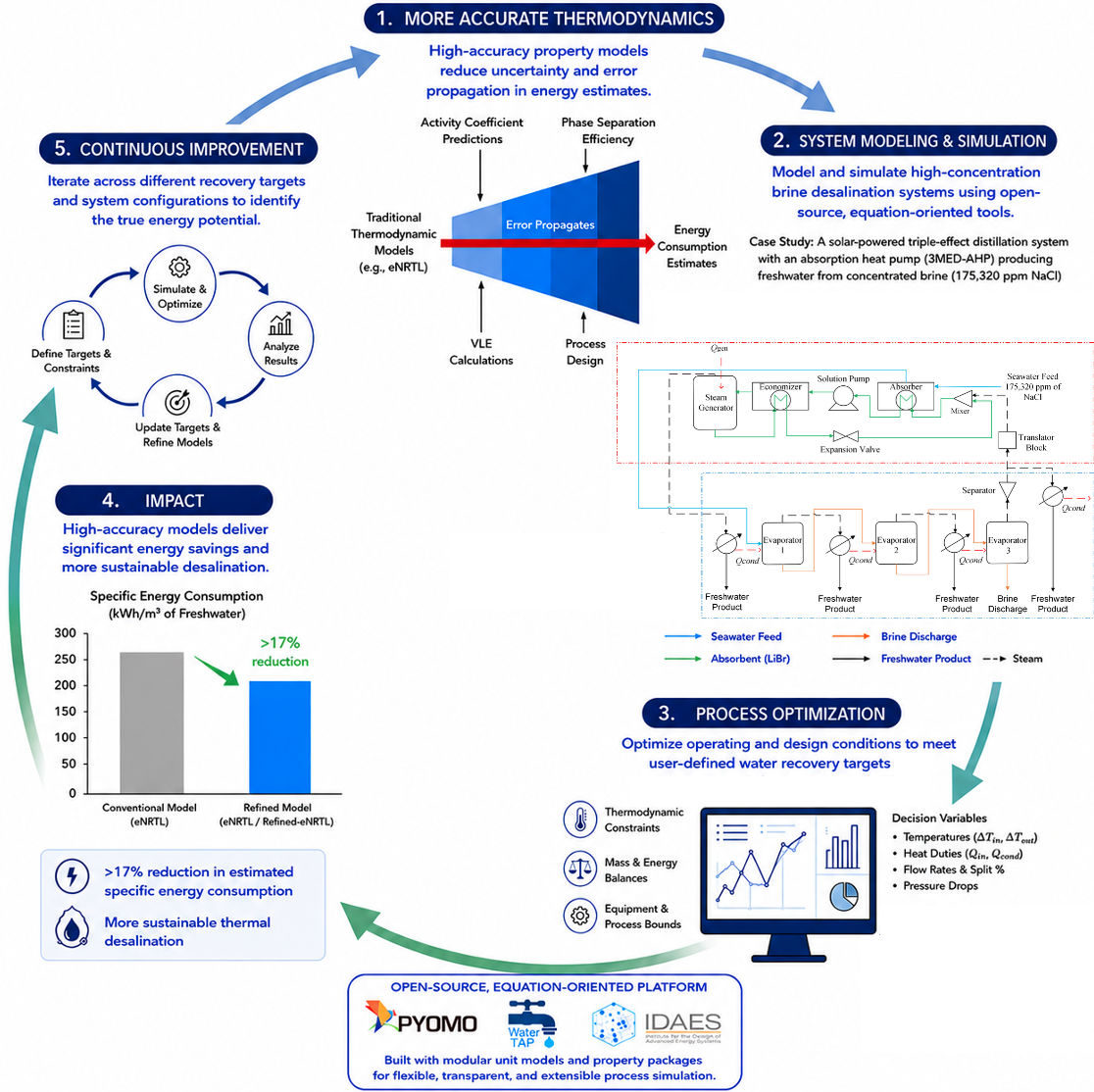
She later contributed to two projects funded by the National Alliance for Water Innovation. One project focused on modeling and simulating high-concentration brine desalination systems using open-source Python-based tools including Pyomo, WaterTAP, and IDAES. Her work demonstrated how the incorporation of more accurate thermodynamic models, which improved predictions of thermodynamic properties, reduced the estimated specific energy consumption of thermal desalination systems by more than 17% compared to conventional methods.
Innovations in Water Treatment and Future Directions
Desalination plays an increasingly important role in addressing global water scarcity, but many processes are highly energy-intensive and involve complex brine chemistries that are difficult to model accurately. In addition to conducting the research, Nazia also developed reusable software packages and documentation that were made accessible to the broader research community through WaterTAP’s open-source GitHub repository.
More recently, she has been developing interpretable models for long-term membrane fouling prediction using ultrafiltration system data. Membrane fouling remains one of the largest operational challenges in membrane-based water treatment due to its dynamic and multifactorial nature. Nazia’s work aims to improve system management and reduce operational costs by creating models that can better predict fouling trends across operational cycles.
Her current research focus explores large language model-based approaches for discovering governing physics in complex systems.
Because her work spans multiple disciplines, adapting to the pace and breadth of the field has been one of the biggest challenges of her PhD journey.
“Coming from an experimental and industrial background, moving into work that involves coding, modeling, and artificial intelligence felt overwhelming at first,” she said. “I found myself learning new tools and ways of thinking while simultaneously tackling new research challenges.”
Despite these hurdles, she sees the interdisciplinary nature of the field as one of its most rewarding aspects. Her work has the potential to benefit researchers, engineers, industry professionals, and policymakers working to improve the efficiency and sustainability of water treatment systems.
Looking ahead, she hopes to continue pursuing research that addresses real-world challenges while also contributing to expanding educational opportunities in developing communities.
“I believe education empowers individuals to create better opportunities for themselves,” she said.
In addition to her research, Nazia recently became the programming director of the NAWI Next Generation Leadership Council, where she helps lead initiatives that connect students, researchers, and industry professionals through events such as technical webinars. After graduation, she hopes to pursue a research-focused role in either national laboratories or industry.
Nazia’s journey reflects both personal determination and a willingness to step into unfamiliar spaces. What began as a decision to challenge herself has evolved into a career dedicated to solving complex problems and building tools that can make a lasting impact.
